Anticorps pour la cytométrie en flux : IREM2/CD303, Kappa, Lambda, F(ab)'2, CD19
Kits Apoptose
Kit d'analyse des cellules souches
Références bibliographiques :
http://www.scielo.br/scielo.php?pid=S0102-86502017000800648&script=sci_arttext
http://www.sciencedirect.com/science/article/pii/S1357272517302339
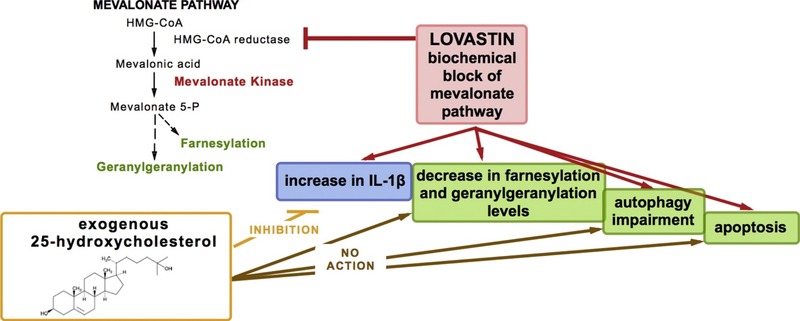
An article published this year in “The International Journal of Biochemistry & Cell Biology" using our “Apoptosis Detection Kit FITC", by our customers from Institute for Maternal and Child Health, Trieste, Italy, in the analysis of how 25-Hydroxycholesterol and inflammation in Lovastatin-deregulated mevalonate pathway. Congrats and Thanks.
Summary :
Mevalonate pathway deregulation has been observed in several diseases, including Mevalonate kinase deficiency (MKD). MKD is a hereditary auto-inflammatory disorder, due to mutations at mevalonate kinase gene (MVK), encoding mevalonate kinase (MK) enzyme. MVK mutations have been reported as associated with impairment of mevalonate pathway with consequent decrease of protein prenylation levels, defective autophagy and increase of IL-1β secretion, followed by cell death. Since 25-hydroxycholesterol (25-HC), a metabolite of cholesterol, can suppress IL-1β production, thus reducing inflammation, we evaluated the effect of 25-HC in an in vitro model of mevalonate pathway alteration, obtained using Lovastatin. Human glioblastoma cell line (U87-MG) was chosen to mimic, at least in part, the central nervous system impairment observed in MKD; 25-HC effects were evaluated aimed at disclosing if this compound could be considered as novel potential drug for MKD.